Thank you to our speakers, sponsors, and delegates who joined us in March for the summit! If you are interested in the 2025 event, please get in touch at info@hansonwade.com
Your Next Event in the Series is:
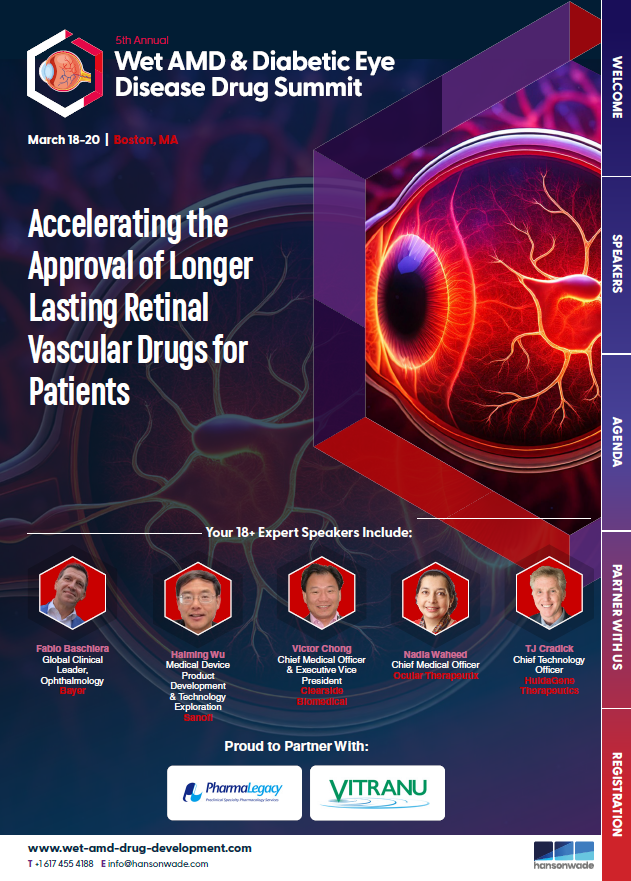
Having trouble downloading the brochure? Let us know and we’ll email it to you instead.
Browse the Full Event Guide to Discover:
18+
industry leading speakers from the likes of Bayer, Sanofi, EyePoint Pharmaceuticals, RevOpsis, HuidaGene Pharmaceuticals, Ocular Therapeutix, and more
6
hours of intimate pre-conference networking and focused discussions identifying effective biomarkers for retinal vascular disorders, exploring innovative clinical trial design, and optimizing clinical endpoints enhance regulatory success
8+
hours of dedicated networking time across speed networking, panels, and interactive Q&A
1
scientific poster session, giving you the opportunity to display your own research to this diverse biopharma audience of wAMD, DR, DME, & RVO drug developers
Exclusive Insights from the Likes Of:
- Fabio Baschiera, Global Clinical Leader, Ophthalmology, Bayer
- Haiming Wu, Medical Device Product Development & Technology Exploration, Sanofi
- Nadia Waheed, Chief Medical Officer, Ocular Therapeutix
- Victor Chong, Chief Medical Officer & Executive Vice President, Clearside Biomedical
- TJ Cradick, Chief Technology Officer, HuidaGene Therapeutics
- Michael lp, Medical Directo, Doheny Image Reading Center
Companies on the program included:



