Your Industry-Dedicated Event for Accelerating Retinal Vascular Drug Approvals
The 2025 Wet AMD & Diabetic Eye Disease Summit was an essential meeting for industry leaders, research innovators, and clinical experts dedicated to transforming the landscape of retinal vascular disease treatments.
The current landscape of wet AMD and diabetic eye disease treatments is evolving with major pharmaceutical players like Novartis, Roche, and Regeneron advancing innovative therapies, including gene therapies and biosimilars. Gene therapies such as RGX-314 are showing promise for wet AMD, while biosimilars like Ranibizumab are improving accessibility and affordability in managing diabetic eye diseases.
Your peers joined from pharma and biotech to delve into comprehensive discussions and presentations that will shape the future of wet AMD and diabetic eye disease management.
Key Features for 2025:
Optimizing Clinical Trial Design: Gain insights from Bayer and Aviceda Therapeutics on structuring trial arms that capture diverse patient populations, assessing drug delivery methods, and evaluating clinical case studies in retinal vascular diseases
Innovative Drug Delivery Methods: Examine advancements led by Ocular Therapeutix and Sanofi that ensure consistent formulation across trial phases, promote patient adherence, and sustain drug efficacy through targeted delivery systems
Biomarker Approaches & Clinical Endpoints: Explore the role of imaging technologies and the validation of surrogate biomarkers, with insights from Opus Genetics and Ocugen, enhancing the ability to predict treatment responses and refine clinical endpoints
Gene Therapy Advances: Discover the latest in gene therapy with HuidaGene and Adverum's innovative candidates, as they push the boundaries of retinal disease treatment through genetic interventions that promise long-term solutions and potential cures
Market Analysis: Gain valuable insights into the retinal disease market, evaluating emerging trends, potential market dynamics, and strategies for successful product positioning from industry leaders
Your Fellow Retinal Vascular Disease Experts Joined to:

Engage with New Voices & Companies: Experience fresh perspectives from new speaking organizations for 2025 including Sanofi, Opus Genetics, Ocugen, EyePoint Pharmaceuticals, Outlook Therapeutics, EOM Pharmaceuticals, RevOpsis, Ocular Therapeutix, Clearside Biomedical and HuidaGene Therapeutics

Exclusive Networking Opportunities: Connect with over 70 global biopharma leaders, including CEOs, clinical researchers, and innovators, through curated networking sessions designed to foster meaningful collaborations and partnerships

Participate in Poster Sessions: Share your own research with peers and gain valuable feedback during dedicated poster presentations, providing a platform for visibility and professional growth

Interactive Workshops: Join workshops that delve into practical tools and strategies, enhancing your knowledge in clinical trial design, patient engagement, and innovative biomarker applications

Explore Market Insights & Opportunities: Stay ahead of industry trends with sessions that provide an in-depth analysis of market shifts, emerging therapeutic opportunities, and strategic directions for future advancements in retinal disease treatments
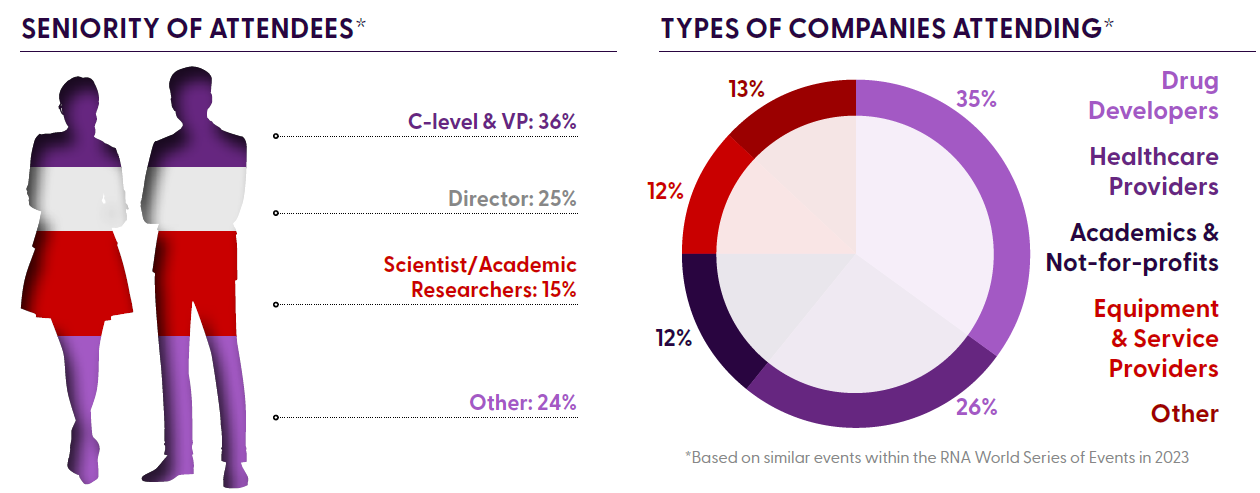
Who Will You Meet?